Chemistry in Community Explain Several Differences Between a Saturated Fat
This answer is not useful. Saturated fasts has a single bond while unsaturated have several bonds between the carbon atoms.

Organic Chemistry What Is The Difference Between Saturated And Unsaturated Fatty Acids Chemistry Stack Exchange
Some unsaturated fats such as coconut oil may become a solid if the room is at a cooler temperature than normal.
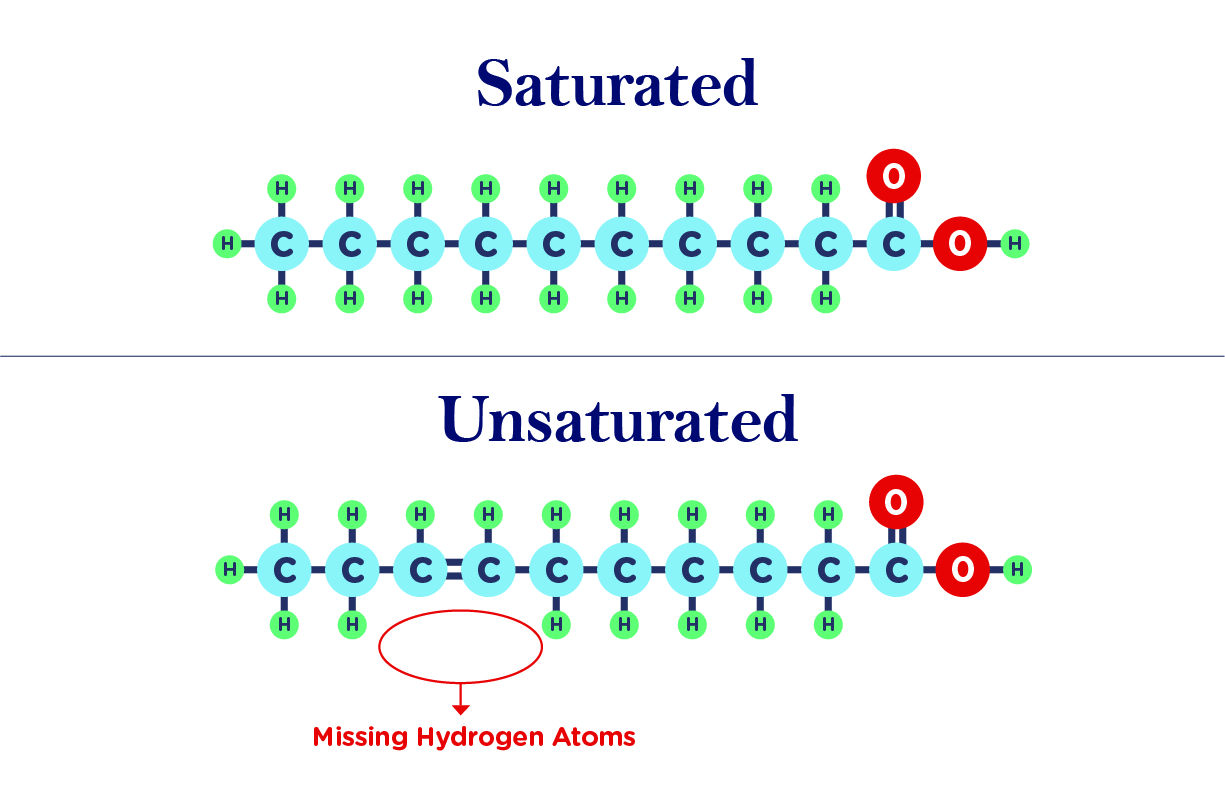
. Saturated fatty acids lack double bonds between the individual carbon atoms while in unsaturated fatty acids there is at least one double bond in the fatty acid chain. This is the main difference between saturated and unsaturated fats. A fat molecule made of saturated fatty acids is saturated fat.
Explain the difference both in terms of structure and in terms of properties between a saturated fat and an unsaturated fat. The major difference between saturated and unsaturated fats is in their bond composition. Saturated fats have the maximum number of hydrogens possible while unsaturated fats have a double bond between two of the carbons.
Now over here or all single bonds between the carbons. Polyunsaturated - many carbons have room for hydrogen. This gives it a straight structure which allows them to stack on top of each other.
The difference between saturated and unsaturated fat lies in the number of double bonds in the fatty acid chain. In saturated fat the carbon atoms are fully saturated with hydrogen atoms and do not contain double bonds in between carbon atoms while the unsaturated fatty acid contains double bonds. Contains at least one double bond.
Saturated fats tend to be in solid form like Crisco and butter. This is the biggest difference between a saturated and unsaturated fatty acid. It is fully saturated with hydrogen atoms.
Differences in chemical structure. The following illustrations explain why. The reason this happens is because of their different chemical structures.
The main difference between fats and oils is the degree of saturation. Fats and oils are two types of essential macronutrients made up of chains of fatty acids. The main source of saturated fat is the animal products and unsaturated fat is the plant products with some exceptions.
Good for consumption but excessive may increase cholesterol. Trans fats are the worst fats as they increase the risk of heart diseases diabetes and strokes. Contains a single bond.
In other words biochemistry is the chemistry of life. Foods like cookies tend to be flat when made with oil. Due to having straight tails saturated fats are dense and solid in nature at room temperature.
The saturated fats will be solid at room temperatures and the unsaturated fats will be liquid. Saturated fasts has a single bond while unsaturated have several bonds between the carbon atoms. In saturated fats each carbon has four hydrogens bonded to it.
Every carbon wants to have four bonds. In addition saturated fats can increase your risk of coronary heart diseases and other metabolic. Now this one of the right and I dont know anything about it it has all single bonds.
But let us find out the other differences between the two. Saturated fat is the type of fat in which fatty acids form single bonds whereas unsaturated fat is the type of fat in which there is one or more double bond in the fatty acid food chain. It studies all the reactions inside and outside of cells.
Saturated fats do not contain a double bond between carbon atoms. This means that the carbon atoms cannot fit anymore hydrogen atoms into the chain. Saturated and unsaturated fats vary greatly in their properties.
There is one caveat to this though. Whereas when made with butter or Crisco they are thicker and fluffier. Both trans fats and saturated fats are bad fats.
Oils are mainly plant-derived and are composed of unsaturated fats. Saturated fats lack double bonds between the individual carbon atoms while in unsaturated fats there is at least one double bond in the fatty acid chain. Fats are solid with high melting point and oils are liquids with low melting points How do you turn an unsaturated oil to a solid.
Arrow_forward What holds the fat molecules in an oil together so that it remains a liquid. Unsaturated fats linoleic acid olive oil and alpha linoleic acid 2 6 Image 4. Show activity on this post.
Chemistry is one of the basic sciences that studies the organic and inorganic substances and interactions of atoms and molecules. In the first illustration seven molecules of completely saturated fats contain only single covalent bonds between the carbon atoms represented as the corners of the zigzag line and each carbon is bound to two hydrogen atoms neither of which is shown in the illustration. When all the carbons are fully bound to hydrogens the fatty acid is said to be saturated.
The key difference between trans fat and saturated fat is that the trans fats contain double bonds between carbon atoms of their fatty acid chains while saturated fats do not contain double bonds. This answer is useful. Excessive consumption leads to heart diseases.
Not to be consumed more than 10 percent of total calories per day. The different types of fatty acids and their corresponding chemical structures. But well talk about that in a second.
Fats are mainly animal-derived and are composed of saturated fats. Whats more saturated fat molecules are solid at room temperature. The major difference between saturated and unsaturated fats is in their bond composition.
React with H2 and a nickel catalyst and a temp of 60 degrees the double bond is removed and the Hydrogen can join onto. Saturated fatty acids are fatty acids with no double bonds and unsaturated fatty acids are fatty acid with one or more double bonds. All the bonds between carbon atoms are single and the hydrocarbon chain has a straight shape.
In contrast an unsaturated fat is a fat compound in which the fatty acids have both single and double bonds. Saturated fats tend to be solid at room temperature and from. Not to be consumed more than 30 percent of total calories per day.
Saturated fats Butter processed meat hydrogenated oil. Biochemistry is and interdisciplinary science that has composed of chemistry and biology. The difference between saturated and unsaturated fat lies in the number of double bonds in the fatty acid chain.
So this is saturated fat because a way to think about it is its saturated with as many hydrogens as possible. Saturated fat is a fat compound in which the fatty acids all have single bonds. There is two types of structural configuration for unsaturated fatty acids due to the.
Unsaturated fats are typically liquid like oils. The worse fats are actually this one right over here. Saturated fats exist as solid at room temperature while unsaturated fats appear as liquid at room temperatures.

Saturated Fats Unsaturated Fats And Trans Fats Video Khan Academy

No comments for "Chemistry in Community Explain Several Differences Between a Saturated Fat"
Post a Comment